
Researchers at Texas University have published a groundbreaking study demonstrating the positive impact of carnitine supplementation on a gene mutation common in ASD.[1] The results specifically advocate taking the amino acid supplement during pregnancy to protect the delicate embryonic neural structures. The research adds to the growing body of evidence that ASD risk and symptoms can mitigated with dietary changes.
Human Energy Production
Virtually every cell in the body contains energy production apparatus called mitochondria – referred to as the ‘powerhouse’ of the cell. The mitochondria take the energy from food and convert it into a form the cell can use.
When you go on abroad you convert money to another currency, like Dollars to Euros. Mitochondria do the same and convert fats, sugars and even proteins into an ‘energy currency’ called ATP (Adenosine Triphosphate). The body cannot store ATP, there is only enough for about 7 seconds, so it must be made continuously to stay alive.
Carnitine and Supplementation
The liver and kidney are responsible for the internal production (biosynthesis) of an amino acid called carnitine from other amino acids in the diet, combined with specific vitamins and minerals. Carnitine is vital to getting fatty acids from the blood across the cell membrane to provide fuel for the mitochondria – also known as a carnitine ‘shuttle’. Carnitine deficiency impairs the body’s ability to transport and use fats which results in muscle weakness and liver problems.
Carnitine supplements increase the rate of fat metabolism, and in some cases are taken to lose weight and also enhance muscle mass. The supplements can also reduce fatigue, boost mental alertness, improve focus and even mood by increasing energy metabolism in the brain. Carnitine therapy has previously been shown to reduce ASD symptoms[2] and improve muscle tone.[3]
Mitochondrial Function
The organs with the greatest energy requirements have the most mitochondria (the brain, gut, muscles and nervous system) which are the same organs and systems typically affected by ASD – inspiring research into the relationship between mitochondrial function and ASD.
Mitochondrial diseases used to be rare, but now represent some of the most common causes of metabolic diseases[4] and have been associated with several other neurological conditions.[5] Other dysfunctions in the mitochondrial ability to process types of fatty acids to release energy have also been linked to ASD.[6]
The exact function of mitochondria in the brain is not completely understood. In addition to providing ATP for energy they also regulate some fundamental aspects of neural function including: triggering cell death; mediating neurotransmitters and the transmission of messages via calcium across nerve synapses – they are also highly sensitive to their environment.[7]
The Changing Brain
Neuroplasticity is the mechanism that allows the brain to change and ‘grow’ throughout life. Many organs in the body cannot renew themselves and scientists used to think the brain was the same, but research has clearly shown otherwise!
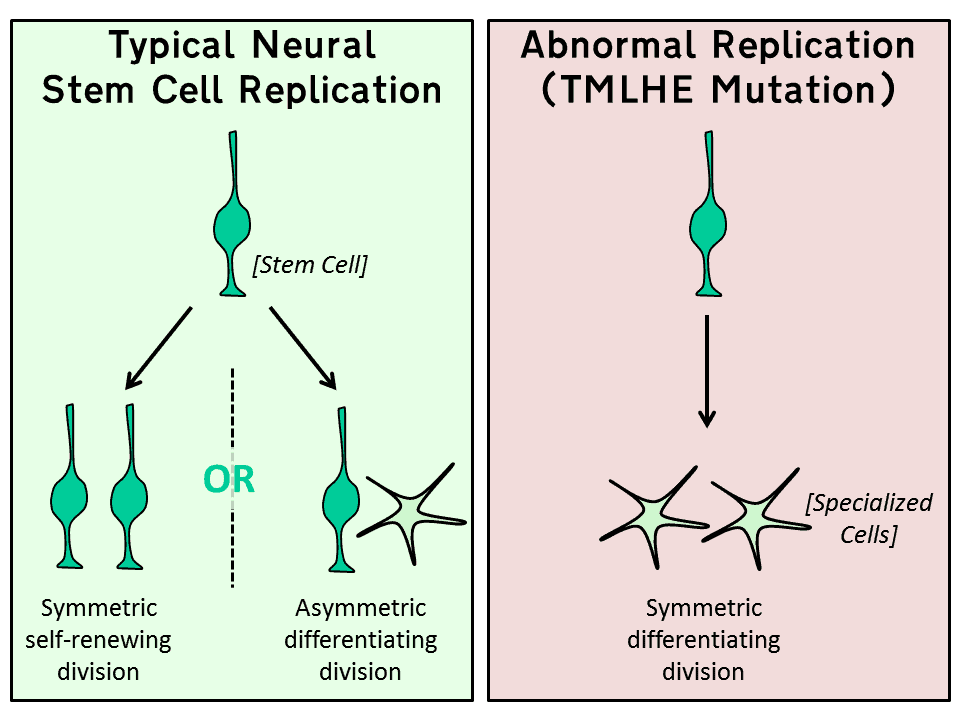
According to this latest research by Texas University the stem cells responsible for growing new neurons are very sensitive to the cellular environment. Healthy stem-cells undergo both self-renewing symmetrical division (producing two stem cells) and asymmetric differentiating division (producing one stem cell and one specialized cell).
Figure 1: Neural Stem Cell Replication and Division
However, when the cell is oxidized (too many free oxygen radicals) this process goes wrong and two specialized cells are produced instead of one stem-cell and one specialized cell.
In essence this means the brain runs low on the stem-cells needed to grow new neurons which impacts the brains growth. During embryonic development it is especially crucial that there are sufficient stem cells to create new structures.
Genetic Mutations
This latest research focused on a faulty gene coding for one of the enzymes involved in carnitine synthesis (trimethyllysine dioxygenase) – the TMLHE gene. Without this gene the body cannot produce carnitine or use fat as a fuel source. When carnitine supplements were given to experimental animals with the same TMLHE mutation, their ability to use fat as a fuel source was restored and at the same time their neural stem cell replication was repaired.
Studies have found the incidence of this specific gene mutation in the general population to be between 1 in 350[8] and 1 in 900[9] (which is relatively high) and it is more common in boys who only have a single copy of the gene, preventing them from synthesizing any carnitine internally. This correlates with the increased incidence of ASD in boys compared to girls (approximately 3:1).
Evidence Summarized
- Defects in the TMLHE gene prevent the production of carnitine (needed to utilize fat for energy)
- If fat cannot be used the body must rely on glucose, which produces more free radicals than fat
- Free radicals cause oxidized cellular environments (usually neutralized with antioxidants)
- Antioxidant levels are lower in ASD individuals so the mitochondrial environment remains toxic
- Toxic mitochondria:
- Cannot produce the energy required for high energy systems (like the gut and brain)
- Cannot support the production of neural stem cells
- Have impaired signaling across neural junctions
- Carnitine supplementation could protect fetal brain development and improve ASD symptoms
Risk Reduction
ASD is a complex condition which is likely a result of many compounding gene mutations or epigenetic regulation issues. Although this research was conducted on mice the strength of the evidence and relevance of the findings is very strong. The high incidence of the TMLHE mutation combined with the low risk and cost of carnitine supplementation makes this a prime candidate to be added to nutritional guidelines for pregnant women in the near future.
Once we understand the science of mitochondria and carnitine some pieces of the ASD puzzle become clearer. ASD could be the result of a vicious cycle of biochemical cellular mechanisms creating toxicity and specifically impacting neural development. Nutritional therapy with carnitine could pave the way to greater understanding, treatment and crucially prevention of a range of neurodevelopmental disorders including ASD.
References:
[1] Zhigang Xiecorrespondenceemail, Albert Jones, Jude T. Deeney, Seong Kwon Hur, Vytas A. Bankaitis. “Inborn Errors of Long-Chain Fatty Acid β-Oxidation Link Neural Stem Cell Self-Renewal to Autism.” Cell Reports Volume 14, Issue 5 (February 2016), p991–999
[2] Mark N. Ziats, Mathew S. Comeaux, Yaping Yang, Fernando Scaglia, Sarah H. Elsea, Qin Sun et al “Improvement of regressive autism symptoms in a child with TMLHE deficiency following carnitine supplementation” American Journal of Medical Genetics, Part A, Volume, 167 Issue 9 (2015), 2162–2167
[3] Geier DA1, Kern JK, Davis G, King PG, Adams JB, Young JL, Geier MR. “A prospective double-blind, randomized clinical trial of levocarnitine to treat autism spectrum disorders.” Medical Science Monitor Volume 14, Issue 6 (June 2011), PI15-PI23
[4] Zeviani M1, Bertagnolio B, Uziel G. “Neurological presentations of mitochondrial diseases.” Journal of Inherited Metabolic Disease Volume 19, Issue 4 (1996), p504-520
[5] Richard E. Frye and Daniel A. Rossignol “Mitochondrial dysfunction can connect the diverse medical symptoms associated with autism spectrum disorders” Pediatric Research Volume 69 (2011), 41R-47R
[6] Clark-Taylor, Tonya, and Benjamin E. Clark-Taylor. “Is autism a disorder of fatty acid metabolism? Possible dysfunction of mitochondrial β-oxidation by long chain acyl-CoA dehydrogenase.” Medical hypotheses 62, no. 6 (2004): 970-975.
[7] Martin Picarda and Bruce S. McEwenb “Mitochondria impact brain function and cognition” Proceedings of the National Academy of Sciences (PNAS USA)Volume 111, Issue 1, 7–8
[8] Patrícia B. S. Celestino-Sopera,Sara Violanteb, Emily L. Crawfordd, Rui Luoe, Anath C. Lionelf et al. “A common X-linked inborn error of carnitine biosynthesis may be a risk factor for nondysmorphic autism” Proceedings of the National Academy of Sciences (PNAS USA)Volume 109, Issue 21, 7974–7981
[9] C Nava, F Lamari, D Héron, C Mignot, A Rastetter, B Keren, D Cohen, A Faudet, D Bouteiller et al. “Analysis of the chromosome X exome in patients with autism spectrum disorders identified novel candidate genes, including TMLHE” Translational Psychiatry, Volume 2 (2012), e179



Is carnitine only a preventive, or could it also be used to treat or lessen autistic symptoms?
Can anyone recommend a some brands of quality supplements that include carnitine, particularly prenatal combinations?